KMU 479
MATERIALS
SCIENCE
Midterm Examination
Instructor:
Dr. Selis Önel
Please give brief answers
to the following questions. All answers should be in English.
Question
1. (12
pts)
One
way of categorizing materials is done as:
1.
Metals and alloys, 2. Ceramics and glasses, 3. Polymers, 4. Semiconductors, 5.Composites
Match
the following properties with the appropriate category by putting M for Metals,
P for Polymers and C for Ceramics next to each property
a) Strong,
ductile
b) Soft,
ductile, low strength, low density
c) Brittle,
glassy, elastic
d) High
thermal & electrical conductivity
e) Thermal
& electrical insulators
f) Non-conducting
(insulators)
g) Opaque,
reflective
h) Optically
translucent or transparent
i) Compounds of metallic and non-metallic elements
j) Metallic
bonding
k) Ionic
bonding
l) Covalent
bonding by sharing of e’s
Question 2. (10 pts)
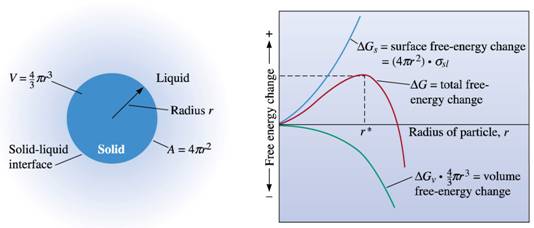
Assume
that during homogeneous nucleation, a spherical interface is created when a
solid forms from the liquid. This solid may either grow into a stable nucleus
or redissolve.
Derive
the equation for the critical nucleus r*, which is the minimum size of a stable
crystal.
According
to this equation, how does supercooling (undercooling)
affect the critical nucleus required for nucleation?
(Note:
Assume that the driving force for solidification, i.e. the free energy per unit
volume, ΔGv is given by ΔHf(ΔT/Tm) where ΔT= Tm –T. Tm is the melting temperature and ΔHf is the latent heat of fusion)
Question 3. (15 pts)
Below,
atomic radius, crystal structure, electronegativity,
and the most common valence are tabulated, for several elements; for those that
are nonmetals, only atomic radii are indicated.
Element ΔR% Structure negativity
Cu FCC 2+
C -44
H -64
O -53
Ag +13 FCC 0 1+
Al +12 FCC -0.4 3+
Co -2
Cr -2
Fe -3
Ni -3 FCC -0.1 2+
Pd +8 FCC +0.3 2+
Pt +9 FCC +0.3 2+
Zn +4
I. Which
of these elements would you expect to form the following with copper and why:
(a) A substitutional solid solution having
complete solubility?
(b) A substitutional solid solution of incomplete
solubility?
(c) An interstitial solid solution?
II.
Suppose 1 atomic % of these elements is added to copper without exceeding the
solubility limit. Which one would be expected to give the higher strength
alloy?
Question 4. (13 pts)
Circle
the correct word in the following sentence or fill in the blank with one of the
words given below:
Composition, Freezing zone, Growth, Liquid,
Microstructure, Nucleation, Solid, Temperature
a)
In materials science and engineering applications, transformation of a to is probably the most important and common
phase transformation.
b)
is the temperature
difference between the liquidus and solidus lines. Within this zone two phases
exist at all times. The amounts of the two phases (the proportion of solid to
liquid) vary, depending on the and .
c)
The two stages involved in the formation of particles of a new phase are:
and . The process involves the formation of normally
very small particles of the new phase(s) which are stable and capable of
continued growth. The stage is simply the increase in size of the
new phase particles.
d)
Three variables that determine the of an alloy are i)
the alloying elements present, ii) the concentrations of these alloying
elements, and iii) the heat treatment of the alloy.
e)
For the condition of phase equilibrium the free energy is a maximum/minimum,
the system is completely stable/metastable meaning that over time the
phase characteristics are constant. For stability/metastability, the system is not at equilibrium, and
there are very slight (and often imperceptible) changes of the phase
characteristics with time.
Question 5. (15 pts)
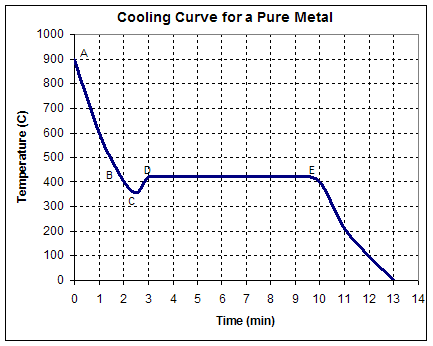
A
cooling curve shows how the temperature of a material varies with time.
Using
the figure above:
(I)
Determine the cooling stages and explain what happens
between indicated points
a) A-B:
b) B-C:
c) C-D:
d) D-E:
e) E - :
(II)
Is this metal well inoculated or not? Why?
(II)
What is the
a)
Pouring temperature?
b)
Solidification temperature?
c)
Superheat?
d)
Cooling rate just before solidification begins?
e)
Total solidification time?
f)
Local solidification time?
g)
Supercooling (undercooling)?
h) Probable
identity of the metal (Si, Zn, Sn or Pb) and why?
Question 6. (15 pts)
In
the Aluminum-Neodymium phase diagram below:

a)
Are any intermetallic compounds present? If so,
identify them and determine whether they are stoichiometric
or nonstoichiometric.
b)
Identify the solid solutions present in the system.
c)
Identify the three-phase reactions by writing down the temperature, the
reaction in equation form, and the name of the reaction.
Question 6. (20 pts)
Consider a Pb–35%
Sn alloy using the phase diagram below

Determine
(a) if the alloy is hypoeutectic or hypereutectic,
(b)
the composition of the first solid to form during
solidification,
(c)
the amounts and compositions of each phase at 184°C,
(d)
the amounts and compositions of each phase at 182°C,
(e) the amounts and compositions of each microconstituent
at 182°C,
(f)
the amounts and compositions of each phase at 25°C.